INTESTINAL ABSORPTION – PERMEABILITY
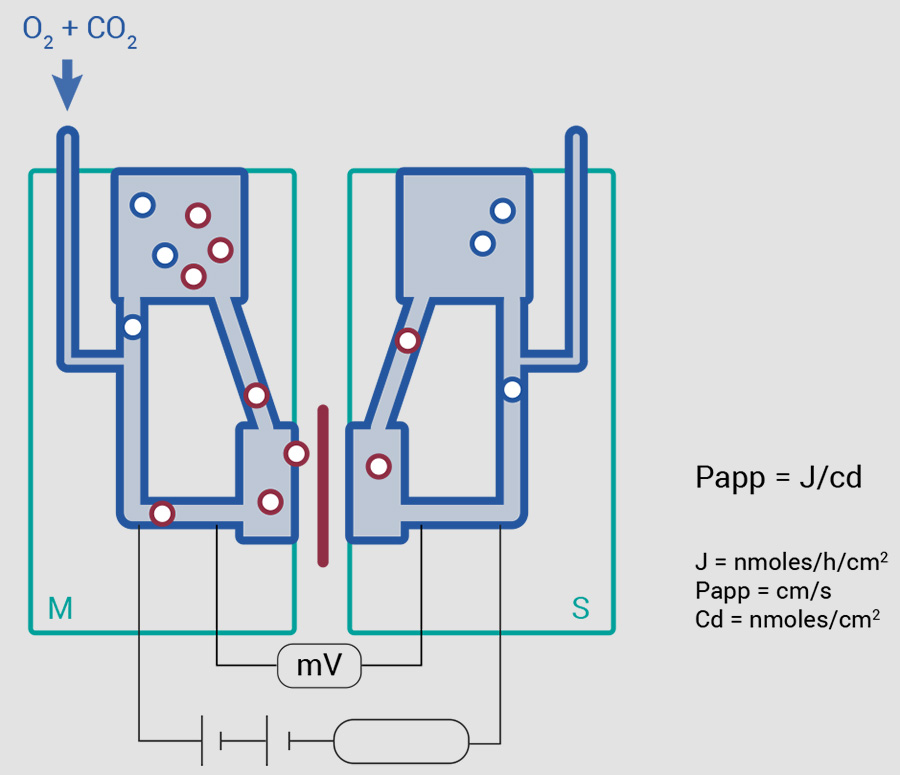
Studies of the intestinal permeation mechanism are carried out using an Ussing chamber.
This device makes it possible to determine permeability, the active ingredient’s rate of transfer, the compound’s impact and physiological factors (composition of the gastro-intestinal environment).
- Permeability studies, type of active-passive transition with increasing doses (3 Log), P influx measurement A-B (apical to basal).
- Specific absorption sites (duodenum, jejunum, ileum, colon).
- Impact of P-glycoprotein: P efflux measurement B-A (basal to apical).
- Impact of excipients.
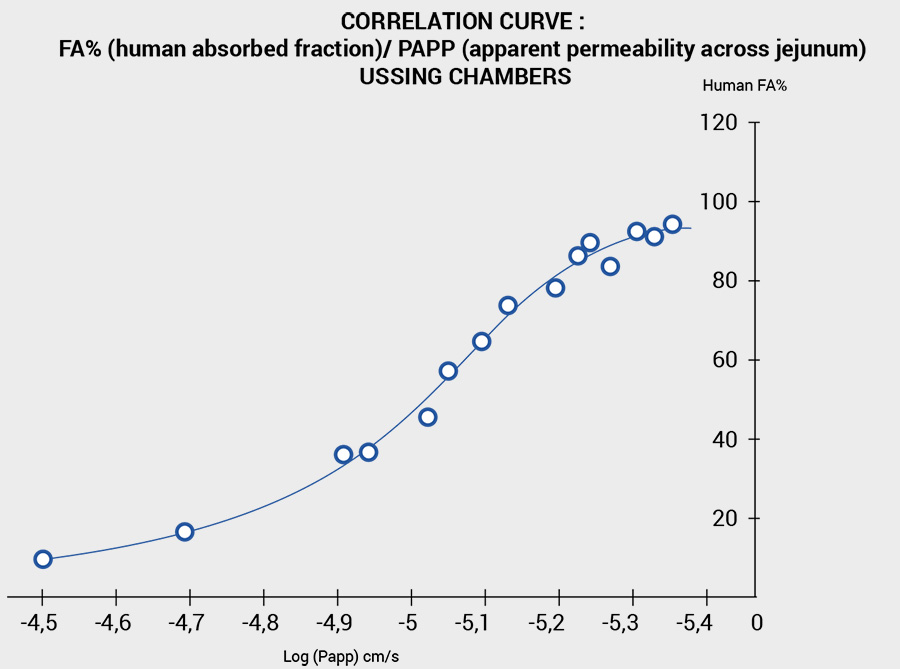
- Fraction absorbed by humans (F%).
Our protocols make it possible to study kinetics following application on the mucosal apical side (A) or the mesenteric serous side (B).
Our protocols also make it possible to precisely determine the permeability of the human intestine.
(Peff: effective permeability), based on the calibration of a series of remedies with variable permeability (providing the Peff and the absorption rate as a percentage, FA%).